CEST
Background
Chemical Exchange Saturation Transfer (CEST) MRI is a new technology that can detect low (millimolar) concentrations of natural non-metallic magnetic marker molecules through the presence of groups with exchangeable protons, such as hydroxyls (OH), amides (NH) and amines (NH2). These protons interact with the water protons detected in MRI through physical exchange, generally referred to in the field as ”chemical exchange”. These protons have a certain resonance frequency, meaning that you get enhancement of sensitivity with high specificity.
Specifik Work
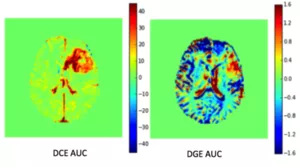
CEST agents are often divided into paramagnetic or diamagnetic agents. Our group’s main research is geared towards diamagnetic agents; either exogenous, as in using natural sugar as a biomarker in tumours (GlucoCEST) (see figure 1 from 7T Lund), or endogenous where we look at the increase of amide protons in tumours (APTw) and the change in concentration of glycosaminogycans (GagCEST) in early osteoarthritis.
Figure 1. An image showing a patient with astrocytoma. To the left is a dynamic contrast enhanced (DCE) area under the curve (AUC) image and to the right is a dynamic glucose enhanced (DGE) AUC image.
Aims
Our specific aims are:
GlucoCEST
To investigate the mechanism of glucoCEST contrast in human volunteers and brain tumour patients and obtaining physiological parameters related to perfusion, permeability and metabolism
To develop, optimize and standardize a clinical MRI (3 Tesla) protocol for reproducible intravenous D-glucose imaging in brain tumours
Expand the current studies to include prostate cancer, first in animal models at 9.4 Tesla and then in patients at 3 Tesla
APTw
To further develop, implement and optimize APTw MRI on our clinical 3 Tesla Siemens scanner.
To evaluate the clinical value of APTw MRI in identifying non-Gd-enhancing high-grade gliomas.
To quantify the accuracy of APTw MRI in distinguishing pseudoprogression from true tumor progression in gliobastomas treated with chemoradiation therapy
GagCEST
To Implement and optimize a gagCEST protocol for use at ultra-high field 7T
To validate the method’s sensitivity to cartilage glycosaminoglycan content using suitable phantoms and excised cartilage samples
To use gagCEST in longitudinal studies in cohorts of patients with osteoarthritis or at risk of developing osteoarthritis. GagCEST will then be valuable tool in our efforts to learn more about the early stages of the disease and how it develops over time.
Collaborators
The research towards our aim is conduced by a network of scientists, both locally in Lund/Malmö and at other sites.
The National and International collaboration sites are:
National:
University Hospital of Linköping - PI Prof. Peter Lundberg
International:
Johns Hopkins University Medical School, Baltimore, US - PI Jinyuan Zhou
F.M. Kirby Research Center, Kennedy Krieger Institute, Baltimore, US - PI Prof. Peter van Zijl
Key References
Xu X, Yadav NN, Knutsson L, Hua J, Kalyani R, Hall E, Laterra J, Blakeley J, Strowd R, Pomper M, Barker P, Chan K, Liu G, McMahon M, Stevens RD, van Zijl PCM. Dynamic Glucose-Enhanced (DGE) MRI: Translation to Human Scanning and First Results in Glioma Patients. Tomography. 2015 Dec;1(2):105-114.
Knutsson L, Xu J, Ahlgren A, van Zijl PCM. Chemical Exchange Saturation Transfer and Arterial Spin Labeling: How Similar Pulse Sequences detect Different Phenomena. Magn Reson Med. 2018 Oct;80(4):1320-1340
Knutsson L, Seidemo A, Rydhög Scherman A, Markenroth Bloch K, Kalyani RR, Andersen M, Sundgren PC, Wirestam R, Helms G, van Zijl PCM, Xu X. Arterial Input Functions and Tissue Response Curves in Dynamic Glucose-Enhanced (DGE) Imaging: Comparison Between glucoCEST and Blood Glucose Sampling in Humans. Tomography. 2018 Dec;4(4):164-171. doi: 10.18383/j.tom.2018.00025
Zhou J, Heo HY, Knutsson L, van Zijl PCM, Jiang S. APT-Weighted MRI: Techniques, Current Neuro Applications, and Challenging Issues. J Magn Reson Imaging. 2019 Jan 20. doi: 10.1002/jmri.26645
Knutsson L, Sundgren P. Clinical Utility of Amid Proton Transfer Imaging in Patients with Brain Tumours. Magnetom Flash. Issue nr 71. 2/2018. 52-55
For more information about our CEST research please contact:
Linda Knutsson, Ph.D.
Professor, Medical Radiation Physics, Lund University, Lund, Sweden
Adjunct Associate Professor, Radiology, Johns Hopkins School of Medicine, Baltimore, US
Work Phone: + 46 46 17 85 47
SE Mobile: +46 76 785 44 24
US Mobile: +1 410 782 0728
e-mail: linda [dot] knutsson [at] med [dot] lu [dot] se (linda[dot]knutsson[at]med[dot]lu[dot]se) LU research portal profile: Linda Knutsson
CEST
Group Leader
Linda Knutsson, Ph.D.
Professor
e-mail: linda [dot] knutsson [at] med [dot] lu [dot] se (linda[dot]knutsson[at]med[dot]lu[dot]se)
LU research portal profile:Linda Knutsson
Seniors
Jonas Svensson, Ph.D.
Associate Professor
e-mail: jonas [dot] svensson [at] med [dot] lu [dot] se (jonas[dot]svensson[at]med[dot]lu[dot]se)
LU research portal profile: Jonas Svensson
Gunther Helms, Ph.D.
Associate Professor
e-mail: gunther [dot] helms [at] med [dot] lu [dot] se (gunther[dot]helms[at]med[dot]lu[dot]se)
LU research portal profile: Gunther Helms
Pernilla Peterson, Ph.D.
e-mail: pernilla [dot] peterson [at] med [dot] lu [dot] se (pernilla[dot]peterson[at]med[dot]lu[dot]se)
LU research portal profile: Pernilla Peterson
Anna Rydhög, Ph.D.
e-mail: anna [dot] rydhog [at] med [dot] lu [dot] se (anna[dot]rydhog[at]med[dot]lu[dot]se)
LU research portal profile: Anna Rydhög
Doctoral Students
Anina Seidemo, M.Sc.
e-mail: anina [dot] seidemo [at] med [dot] lu [dot] se (anina[dot]seidemo[at]med[dot]lu[dot]se)
LU research portal profile: Anina Seidemo
Research Assistants
Patrick Michael Lehmann, M.Sc.
e-mail:
patrickmichael [dot] lehmann [at] gmail [dot] com (patrickmichael[dot]lehmann[at]gmail[dot]com)